|
Back to Blog
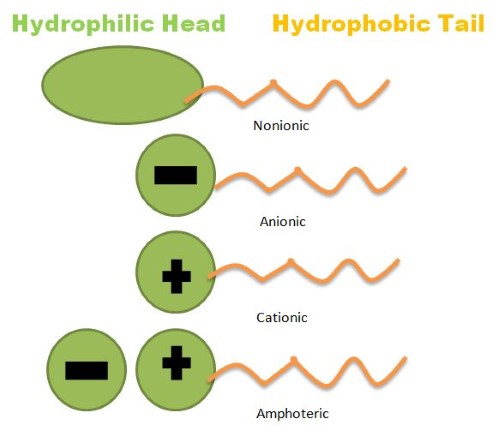
Surfactants, as a rule are substances that act on interfaces or surfaces and can alter their properties. Interfaces are the interface of two different phases, typically liquid solid, gas, or liquid. Surfactants can decrease the tension on the surface of a liquid which makes it more likely to spread across an area. They can also reduce the tension between two different liquids, making it easier to mix. What are surfactants? How do surfactants work?The most commonly used use for nonionic surfactant suppliers is in detergents employed at home. They take dirt off clothes, skin and various other surfaces. The word itself is derived from the word "Surface Active Agent'. Surfactants can modify the properties of surfaces and interfaces by being active on the surfaces they are on. This is why it's important to first understand that an interface can be described as the intersection of two different phases, usually liquid, solid, or gas. Surfactants can decrease the tension on the surface of liquids making it more likely be spread over a surface, or reduce tension between two incompatible liquids making them more easy to mix. Your application will determine the kind of surfactant that you require. There are a variety of surfactants that can be used for different purposes. What are the different types of Surfactant? Based on the purpose for which you are using it you may require one of several types of surfactant that are thickened, pH, high foam, substantive or high foam. Let's look at some typical surfactant examples, kinds of surfactants and their use. Surfactants anionic These surfactants that contain anionic (negatively charged anionic ions) are insoluble at low temperature levels and have hydrophobic anion effects when they are dissolved in water that is warmer. They are capable of attacking all kinds of soils, so are excellent for use in the home. IRO Surfactant are commonly used in the manufacturing of detergent and cleaning products. They are also utilized in other industries like construction, agriculture, and textiles for uses like dyeing, bleaching, demulsifying, corrosion prevention and as an additive to fuels. Non-ionic surfactant It is a surfactant that does not ionize when it is dissolving in water. They are, therefore, extremely stable and exhibit a very limited reaction to acids as well as strong electrolytes. About 50% of surfactant production is comprised of non-ionic surfactants. They possess better emulsifying properties than anionic surfactants and are therefore ideal for the removal of organic and oily dirt. They can be present in shampoos (thanks their capability to assist in emulsification) as well as in perfumes, and also in beauty products like make-up, for instance, they can aid in dispersing the pigment on the skin. Cationic Surfactants While they are not as common than other surfactants used in manufacturing, cationic surfactants can be employed to create fabric softeners. They are ideal because they are well-integrated with anionic surfactants which help remove dirt and staining. One of the most popular surfactants that are cationic is benzalkonium chloride (BAC) that is utilized as a preservative in eye drops and for antiseptic purposes in higher concentrations. Amphoteric Surfactant These surfactants are a combination of anionic and cationic surfactants. These surfactants are relatively fresh and still under development. They have shown numerous positive characteristics throughout the years, such as: Biodegradability Resistance to hard water As an emulsifier These properties, along with other properties These properties, along with many others, create IRO Surfactant appropriate for shampoos, dyes or pharmaceuticals. Airedale Chemical is a top surfactants supplier and manufacturer. As specialists in chemical manufacturing and supply We are ideally placed to provide you with existing surfactants but help you with the development and research of new bespoke solutions. We invite you to explore our collection of Surfactants which provides appearance, description and application information for each.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
